Research at the Laboratory of Crystallography
The Maximum Entropy Method in Crystallography
The Maximum Entropy Method (MEM) is a method to derive the most probably map given a set of experimental data. In crystallography the MEM can be used to determine the electron density in the unit cell that provides the best fit to the scattering data. It is a model-independent approach in contrast to structure refinements, in which the positions of spherical atoms are determined. Therefore the MEM is most suited to study aspects of structures that go beyond the independent atom approximation, like partially ordered structures, the electron density in the chemical bond, and the effects of anharmonic atomic vibrations, as well as the study of modulation functions in aperiodic crystals.
At the laboratory of crystallography we develop a computer program BAYMEM for the application of the MEM to crystallographic problems [1].
Case Studies:
Anharmonic displacements in Cs2HgCl4 as precursor for the phase transitions in this compound (see the figure below) [2,3].
Molecular disorder in relation to packing principles and stability in the organometallic compound LiCp* [4].
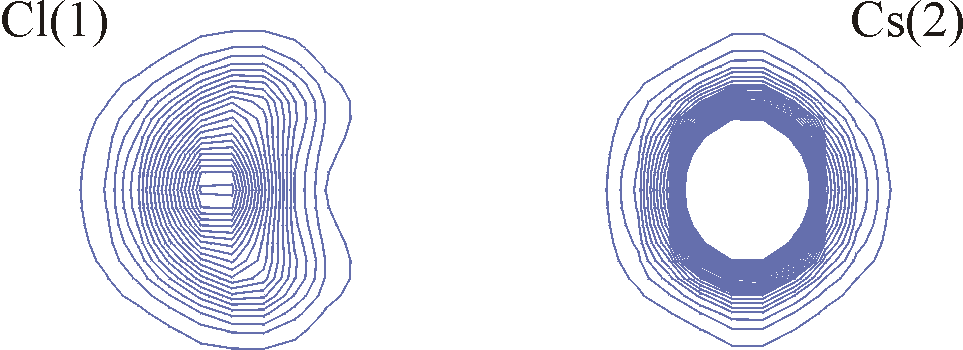
Contour lines of equal electron density of a section of the structure of Cs2HgCl4 going through the atoms Cl(1) and Cs(2). The anharmonic contributions to the displacements of Cl(1) are clearly shown by the non-convex shape of the outermost contour lines.
Publications about this topic:
[1] S. van Smaalen, L. Palatinus and M. Schneider, Acta Crystallogr. 59, 459 - 469 (2003).
[2] B. Bagautdinov, J. Luedecke, M. Schneider and S. van Smaalen (1998): Disorder in Cs2HgCl4 studied by the Maximum Entropy Method. Acta Crystallogr. B 54, 626.
[3] B. Bagautdinov, K. Pilz, J. Luedecke, and S. van Smaalen (1999): Modulated structures of Cs2HgCl4: the 5a-superstructure at 185 K and the 3c-superstructure at 176 K. Acta Crystallogr. B.
[4] R. E. Dinnebier, M. Schneider, S. van Smaalen, F. Olbrich and U. Behrens (1999): Disorder determined by high resolution powder diffraction: The structure of pentamethylcyclopentadienyllithium. Acta Crystallogr. B 55, 35.
